Optical
Emission Spectrometer is an instrument measures the intensity
of several spectral lines simultaneously in the light emitted by the sample,
when the atoms that compose it are exited by an external energy source. The
analysed light is located approximately into the 150nm to 800nm wavelength
range. The whole measuring system is therefore based on the physical phenomenon
that is summarised as follows:
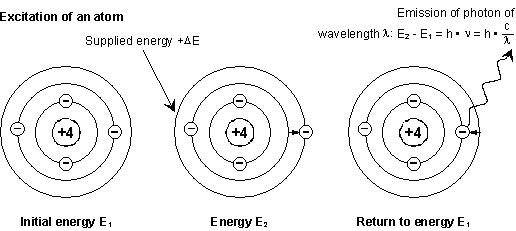
When certain energy is applied to an
atom, some of its electrons change their orbit. When these electrons return to
their initial orbit, a precise energy is restored in the form of a light at a
determined wavelength. This is an atomic phenomenon, and consequently it is
practically unaffected by the chemical or crystalline form of the atom. This
means the instrument can determine, for example the quantity of silicon in
steel; but will not give information about the form under this silicon is to be
found. The following figure gives a rough representation of this excitation.
A sample containing several different
elements will therefore produce light composed of wavelength specific to each
of the elements. By separating these wavelengths by a dispersion system, the
spectrometer can determine which elements are present, the intensity of each of
these wavelengths being a function of the concentration of the considered
element. By measuring this luminous intensity (with a photomultiplier) and by
processing this information with a computer, the instrument can thus determine
the concentration of the considered element.